This article discusses the Medical Store at central-level stores and at regional district-level stores that are independent at health facilities. This supply model represents the tradition top-down system that is operated by governmental or parastatal entity or a nongovernmental organization; however, a number of countries have now decentralized many of their pharmaceutical sector operations, including stores management.
In addition, many supply systems contract out certain operations to private-sector companies. Managing requires skills such as defining and monitoring performance indicators and negotiating payment and service terms. The primary purpose of a central supply store is to receive, hold, and dispatch stock.
Table of Contents
Information for Materials Management
Management of Medical Store’s goal are to protect stored items from lost, damage, theft, or wastage and to manage the reliable movement of supplies from source to user in the most economical and expeditious ways. Effective management of information is the key to achieving these goals. A fully developed system has three key components: an inventory control system; a warehouse management system; and a performance monitoring system. Information is essential to management, collecting, processing, and disseminating information system should be balanced against the costs of ineffective inventory control.
Manual versus Computerized Systems
The information system must include data on products entering and leaving the warehouse, products in stock, products on order, and ways to monitor the progress of order in the supply pipeline. The procurement process involves medical stores input all levels of the distribution system. Information about supplier performance should be compiled throughout the procurement cycle to assist in choosing future suppliers.
Stock records
Contain information about suppliers, customers, prices, stock receipts, stock issues, stock losses, and stock balances. Many medical stores information systems also keep general information on facility location, facility status, population served, method of distribution, delivery schedule, value of issues, and extent of current budget utilization.
Performance monitoring and reporting
A store should monitor and evaluate its operations to identify problems in the system that need to be addressed. Record-keeping and reporting systems should be designed to make the collection of data for routine monitoring as simple as possible.The medical stores must maintain effective communications with suppliers, with procurement and inventory control units, with lower-level warehouses and health facilities, and with managers from the ministry of health.

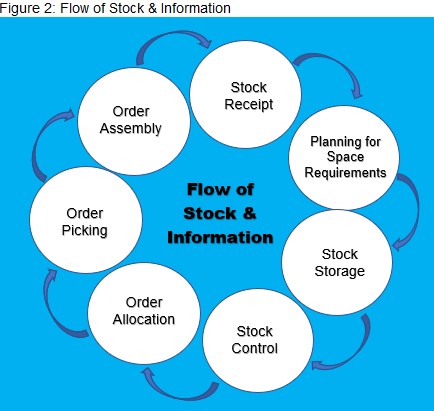
Flow of Stock and Information
Formal and informal physical stock counts should be taken regularly to ensure that the stock is being correctly handled and that losses an inaccuracy is properly accounted for.
The procedure outlined here assumes that those tasks are carried out by an import unit attached to the Central Supply Store (CSS).
Planning for space requirements
Store therefore need to work carefully with their clients to communicate about scale-up of initiatives that will affect space and distribution planning and to develop policies on managing strategic stock. After incoming stock has been checked and approved, it is formally released from the receiving area and moved to the warehouse to be stored in the appropriate zone.
Stock control Using FIFO and FEFO
In order to avoid accumulation of expired and absolute stock, items should be stored and issued on a FIFO or first-expiry/ first-out (FEFO) basis, according to the following guidelines.
a. Order allocation
Under a full distribution system, lower-level stores and health facilities submit requests, or requisition, for supplies.
b. Order Picking
The allocated quantities from the order picking list. This is passed to the store keeper.
c. Order assembly
At a secured shipping location, the supplies are arranged in the order in which they appear on the picking list or requisition voucher.
d. Order dispatched and delivery
In the most common used in-house delivery, supplies are generally distributed according to a fixed delivery schedule.
e. Inventory taking
The supply stores unit must regularly take physical counts of stock that the stock balance on perpetual inventory records is correct.
Zoning Stock with the Store
Medicines and essential medical supplies must have located in a part of the store with the correct combination of temperature and security. In hot climates, it is necessary to store many times in air-conditioned rooms. In humid climates, dehumidifiers are useful for preventing moisture damage. In cold climates, stores may need to be heated in winter to protect products that are damaged by freezing.
a. Cold Storage
the potency of vaccines, sera, test kits, and many other items depends on cold storage. Vaccines, in particular, are temperature-sensitive and must be kept at precisely controlled temperatures from the point of manufacture to the point of administration.
b. Secure storage
Narcotics and other controlled substances should be kept in a secure room or in a safe.
c. Flammable
Such as alcohol and ether, must be stored in special buildings or rooms. The flammables store must be well ventilated and fireproof.

Stock Location within a Zone
Within each zone, stock may be located in fixed, fluid, or semifluid locations. Fixed location systems are the simplest to manage, because each stock item is always stored in the same place, but they waste space.
a. Fixed location system
Each stock item is allocated to specific shelves, pallet racking, or an area of floor.
b. Fluid location system
The store is divided into many designated locations. Each location is assigned a code. Individual items are stored wherever space is available at the time of delivery.
c. Semifluid location system
The combination of the fixed and fluid system, each item is assigned a fixed space for picking stocks.
The full content is only visible to SIPMM members
Already a member? Please Login to continue reading.
References:
Battersby, A., and A. Garnett. (1993). How to Estimate Warehouse Space for Drugs. Geneva: World Health Organization.
Dalberg Global Development Advisors and the MIT-Zaragosa International Logistics Program. (2008). The Private Sector’s Role in Health Supply Chain: Review of the Role and Potential for Private Sector Engagement in Developing Country Health Supply Chains. New York: The Rockfeller Foundation.
Ghiani, G., G. Laporte, and R. Musmanno. (2004). Introduction to Logistics Systems Planning and Control. West Sussex, England: John Wiley & Sons.
John Snow Inc./DELIVER. (2005). Guidelines for Warehousing Health Commodities. Arlington, Va.: John Snow, Inc./DELIVER, for the U.S. Agency for International Development.
John Snow, Inc./DELIVER in collaboration with the World Health Organization. (2003). Guidelines. Arlington, Va.: John Snow, Inc./DELIVER, for the U.S. Agency for International Development.
Mulcahy, D. (1994). Warehouse Distribution and Operations Hand Book. New York: McGraw-Hill Professional. WHO (World Health Organization). 2009. WHO Expert Committee on Specifications for Pharmaceutical Preparations. 43rd Report. Geneva: WHO. 227. WHO New Product Information Sheets (PIS). Since 2000 Edition.

